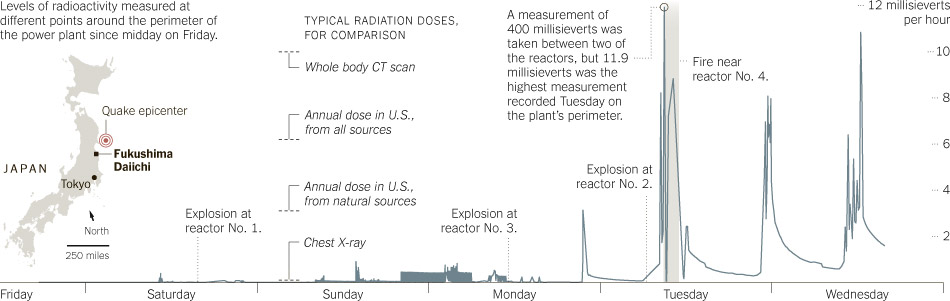
Much recent news and many questions directed at the blog have centered around the detection of plutonium in soil and water surrounding the Fukushima reactors. This post will outline our current knowledge of the situation, as well as potential impacts on the environment and on human health.
Measurements to date
On March 21 and 22, five soil samples around the site indicated the presence of isotopes of plutonium. Two of these samples contained the isotope Pu-238. Typically, a large ratio of Pu-238 to the other isotopes indicates that the material has been produced in a reactor. This is because the production of Pu-238 requires one of these processes to take place:
- Successive neutron captures in U-235 produce U-237. U-237 decays to Np-237, with a half-life of 6.75 days. Np-238 captures another neutron and decays to Pu-238.
- A fast moving neutron causes a Pu-239 nucleus to eject an additional neutron.
This second reaction can take place during the detonation of a nuclear weapon, but is rare. The first is next to impossible in a nuclear detonation, as the weapon blows itself apart before the necessary series of captures and decays can occur. This is why the detection of Pu-238 at two sites indicates that material at those sites came from a reactor. There is not sufficient information at this time to determine whether the material originated from the MOX-fueled unit 3, or from one of the other cores.
Because Pu-238 was not detected at the other three sites, it is thought that the plutonium at those sites is a remnant of past nuclear weapons tests. For reference, the natural rate of plutonium decay in Fukushima City is 0.61 Bq/kg, or 0.61 decays per second in each kilogram of soil. The quantities being measured on the reactor site are at roughly double this same level, according to TEPCO.
Transport Pathways
As stated, the pathway which was taken by the plutonium to the soil of the reactor site is not clear at the present. However, it is generally transported via one of two pathways:
- Adhesion to particulate matter, like smoke.
- Solution or suspension in water.
Whichever route led to the disposition of plutonium on the reactor site, it would be difficult for such plutonium to be transported over great distances. Its high mass means that it is not easily aerosolized, even by fire. Mention has been made of the fact that plutonium will, under the right conditions, burn. However, this burning occurs during plutonium metal’s conversion to plutonium oxide. As the plutonium within each reactor is already in an oxide form, it has no such tendency to burn. Finally, plutonium is not very water-soluble. Under optimal conditions, the solubility of plutonium metal in water is around 55 microgram/L. The solubility of plutonium oxide is even lower.
Could plutonium be transported away from the reactor site, under the current conditions? Potentially, yes. However, it would likely be in minute quantities that have no impact on human health.
Impact of Plutonium on Human Health
As a radiation hazard, plutonium is a danger when ingested or inhaled. This is because it’s an alpha emitter. Alpha particles, while they can be stopped by the skin or a sheet of paper, can severely injure very delicate structures of the body, such as the alveoli in the lungs, or the lining of the gastrointestinal tract. Plutonium is a bone-seeker, but is not efficiently absorbed by the body because of its low solubility in the body’s fluids. The vast majority of ingested plutonium (greater than 99%) is excreted within a week of ingestion. Between 5% and 60% (estimates by different agencies vary) of inhaled plutonium stays within the body, with the rest being exhaled immediately.
In addition, plutonium is chemically toxic like other heavy metals. A number of estimates have been circulated regarding how much plutonium is fatal to humans, many of which have no evidence to support them. Experiments using lab rats have indicated that 50% of those rats die within a month after injection of 700-1000 micrograms of plutonium per kilogram of body weight. This would translate to 47.7-68.2 mg of plutonium injected into a 150-pound person. Since the efficiency of plutonium uptake by inhalation or ingestion is low, the dose needed to actually cause illness or death would be correspondingly higher.
It’s unknown whether the results of experiments on rats translate directly to human exposures. No human has ever died from acute uptake of plutonium. Our information on the health effects of plutonium on humans is derived from the case studies of plutonium workers, who sustain very low doses over a period of decades; a series of studies on chronically ill patients; and the histories of atomic bomb survivors, whose doses are confounded by exposures to a whole host of other radioactive isotopes.
Sources: Journal of Radiological Protection, Los Alamos National Laboratory, TEPCO, American Chemical Society.

 A helicopter shot of Fukushima Daiichi 3 earlier today
A helicopter shot of Fukushima Daiichi 3 earlier today